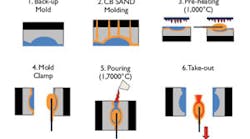
The Convert Mold Process, using the Convert Binder for the production of thin-walled steel castings 4,5,7, was introduced in 1994 (see Fig. 1), and uses a secondary binder that converts organic-bonded molds and cores into a ceramic capable of withstanding high temperatures. Also, it allows castings to be poured while the mold assembly is at elevated temperatures, i.e. 1,000°C.
Initially, there were problems associated with the requirement of immersing the organic bonded mold in the inorganic binder that, along with the high cost, restricted market acceptance of the process. To eliminate these problems, we have developed the Convert Binder-coated sand and the pre-heated burnout cycle for high mold temperatures. The simplified and lower-cost process has been developed to produce steel castings with wall thicknesses in the range of 2.0 mm or less.
For automotive castings, conversion from cast iron to steel has been limited when thin-wall and hollow structures are desired. Automakers’ desire to use steel, because of its high tensile strength and impact resistance, has been restricted primarily by casting process limitations. Also, there are difficulties with high-volume thin-wall casting production, and shrinkage issues at solidification. Issues associated with the nil-point, which loses rigidity and moves toward zero immediately after solidification, cause hot tears and cracks that must be repaired or rejected.
The original Convert Process5, 7 for thin-wall steel casting technology used conventional organic bonded sand molds and cores that were dipped into a proprietary, patented solution called the “Convert Binder.” After dipping the core or mold in the Convert Binder, the mold assembly was dried by evaporation and baked in a conventional oven to remove all of the solvent. The process was adversely affected by the difficulties of handling the binder and the need to remove all of the solvent through evaporation and baking the mold in a conventional foundry oven. Recent improvements to the Convert Binder-coated sand process have minimized material consumption and the pre-heat cycle for high-temperature molds, and reduce the process time from hours to three-minute cycle times, or less.
Development of the hightemperature mold process
It is well documented that cast steel is prone to hot tears and cracking due to the nil point and restrictions created by the molding materials. In investment casting, pre-heating the mold at 1,000°C is used to avoid this problem2, but the investment mold requires long heat cycles in the kiln. The amount of time required to prepare the investment mold for casting is directly proportional to the weight of the mold and mold section size. The larger or heavier the mold, the longer will be the time required in the kiln.
The recent improvements to the Convert Mold Process include using back-up molds to minimize material requirements, reduce cost, and shorten the time required to elevate the coated molds to 1,000°C. In addition to the process changes, a new system for pre-heating the mold cavity efficiently reduces the pre-heating cycle to three minutes or less.
Characteristics of the Convert Binder
The Convert Binder is a complex solution of silane alkoxide and sodium alkoxide. This complex is crystallized using the sodium (Na) ion as its nuclei during the solvent evaporation step when the Convert resin coats the raw sand surface.
Once coated with the inorganic binder, commercially available organic resins are used to produce bonded molds and cores. Convert Binder coated sand has produced excellent casting results using both shell resins and amine-cured polyurethanes systems. These organic binders form organic silicate compounds at 200°C by a catalytic reaction of the Na ion, and later are converted to sand/ceramic-mold media at high temperature. (See Fig. 2.)
The Na ion is known for its high reactivity and reducibility in catalyzing chemical reactions. However, its high reactivity in air and storage difficulties has always presented a safety and handling problem. During the development of the Convert Binder, it was discovered that a silica compound containing the Na ion was produced, which maintains a very stable, high-Na ion whose reactivity can be used in the range from ambient temperature to the boiling point. The Convert Binder is therefore a complex mixture of metal alkoxide and sodium alcholate.1, 2
The Na ion acts as a cross-coupling agent to form organic silica compounds from the organic compounds and silica- containing materials at 200°C. (See Fig. 3.)
The organic silica compound formation was observed by placing phenolic resin on an aluminum plate that had been coated with a film of Convert Binder. The phenolic resin in contact with the Convert Binder spread rapidly when heated to 200°C, demonstrating the high reactivity of the Convert Binder. No change was observed of the phenol resin located directly on the aluminum plate.
Convert Binder coated sand
The major development of the new process is to apply the Convert Binder to the sand prior to the application of organic systems. This change results in a significant reduction in Convert Binder consumption and allows for better adherence since the inorganic is in direct contact with the sand surface, allowing the silica content of the sand to react with the binder.
The Convert Binder reacts successfully with both the polymeric inorganic materials and the organic silica compound to form a new, high-temperature investment-type binder. In addition, the Convert Binder reacts with low-molecular weight olefinic hydrocarbons. Lowolefinic hydrocarbons exhibit high levels of wetability in a wide range of low temperatures, which are conducive to forming highly desirable thin films in the conversion to hybrid inorganic binders.
The Convert Mold is produced using Convert Binder coated sand. (See Fig. 4.) The target strength is above the 0.3 Mpa of thermal deflection strength at 100°C in 60 minutes. The 10 10 50- mm test specimens were prepared using Cera-beads™ (40% Al2O3-60% SiO2) as an aggregate.
The specimens were prepared to compare a phenol resin at 2.2 w%. The specimen was heated by 200°C for 60 minutes and 1,000°C for 60 minutes. The converted organic mold prepared using the Convert Binder coated sand showed acceptable strength for casting at 1,000 °C.
Further testing exposed the Convert Mold to various temperatures to measure strength changes. The samples were exposed to elevated temperatures for 60 minutes at 200°C, 300°C, 350°C, and 400°C. (See Table 1.) At 400°C, a loss of strength was confirmed. While the Na ion forms an organic silica compound between silica and the organic binder at 200°C, the reactive Na ion destroys the benzene ring compounds at 400°C. At 800°C, the mineralization reaction causes a tridymite conversion of silica due to the Na ion contained in the Convert Binder.
Although the Convert Binder results from a hybrid product of organic and inorganic chemistry, it may be difficult to understand since some of the organic or inorganic materials are not currently used conventionally in the iron, steel, and investment casting sectors.
Therefore, what follows is a summary that will help readers to understand the Convert Process and be inspired to use it as a new process alternative for thinwall steel casting.
Historically, the inorganic silicate binders used for casting are silica gel, sodium silicate, and ethyl silicates that form amorphous films. The Convert Binder is an entirely different material than Sol-Gel derived from sodium silicates and ethyl silicates. In the Convert Binder, the Na and silica exist independently as a Na ion in a silica crystal structure. This unique molecule maintains the reactivity of metallic sodium from ambient temperatures to its boiling point.1
The Convert Binder is produced by capturing the Na ion as its nucleus, without oxidation at 67°C, with a ratio of Na:SiO2 = 0.3~1.0. The reactive Na ion is captured in the silica crystal cavity where it retains its reactivity as metallic sodium.
The sodium-silica compound is transformed to C-tridymite via crystobalite at temperatures higher than 800°C, using Na2O derived from the reduction of silica by sodium as a mineral source.1,8 These unique properties of the Convert Binder result in performance that is completely different from that of the Sol-Gel method derived using ethyl silicate in the investment casting process.
Na ion as a reaction catalyst
The function of the Na ion in the Convert Binder has a variety of features relative to the temperature. Convert Binder maintains Na ion reactivity from the ambient temperature to the boiling point. (However, Na ion detection cannot be done by electronic stimulation using an electron microscope.)
The Na ion destroys benzene rings in aromatic compounds by acting as a decoupling reagent at 400°C or lower. Using the Convert Binder-impregnated resin mold eliminates the black smoke and phenol resin burning odors that are typicl during casting with organic binders.
TG-DTA analysis and EPMA analysis for residuals were conducted at 1,000°C. TG-DTA analysis using biomass wood chips containing high levels of aromatic compounds have proven that the destruction of benzene rings were initiated by Na ion in Convert Binder. Untreated wood chips verify the difference between treated and untreated.
Organic silica compounds are transformed to tridymite at 900°C via the intermediate crystobalite by catalytic reaction of Na ion, while quartz is transferred to tridymite only with Na2O. Na2O is formed by reduction of silica with Na in a sodium-silica compound after decomposition of organic silica compounds.1,8
Pre-heating the mold
The process of heating a mold to 1,000°C may prove to be difficult using conventional equipment. At present, most foundries use gas furnaces with high thermal capacities. In addition, the internal spaces within the mold become insulating and reduce the effectiveness of the kiln.
During commercialization a cooperative development was conducted with Tokyo Gas Engineering that resulted in a method to reduce the time required to raise the mold temperature to 1,000°C in 3 minutes or less.
A mold-heating device (see Fig. 5) was built for a surface mold area of 600 600 mm. The heat capacity is 400,000 cal / time. The fuel is a combination of usual gas air and gas oxygen. Heating the mold cavities in the direct, pre-heating position makes it possible to clamp the mold, simplifying the process, which results in cycle times in 3 minutes or less.
A back-up mold was used to minimize the amount of material used in the casting process. The back-up mold material uses inorganic materials, such as refractories with high heat retention like the Thermal Bonding System™ (TBS) of ceramic fibers developed by Japan’s Isolite Insulating Products Co. Ltd.
Cast iron is used to clamp the mold halves together for pouring.
In recent years the use of low heat-expansion sand has been increasing for thinwall casting, due to the demand for thinner casting sections. Conventional silica sand has been used for this process. The thermal expansion of the silica sand is about 2%. This is possible when the temperature is closely controlled while heating. Heating and holding to 570°C controls the thermal expansion, avoiding mold cracking that occurs by the sudden heating to 1,000°C.
Using the Convert Mold Process and reclamation of artificial sand becomes economical, but not required, since the requirement for make-up sand in most units is less than 3%. Ceramic molds treated with the Convert Binders are easy to remove from the casting and to reclaim efficiently. Thin-wall steel castings High-temperature molds are heated to 1,000°C for thin-wall steel casting production. (Note that the melting point of lowcarbon steel is high and it solidifies quickly because of the often-narrow zone between liquidus and solidus.) Cross-sections of 1.6 mm are routinely produced using SAE1010 low-carbon steel. When casting into a mold at ambient temperatures, metal flow is restricted due to temperature loss to the mold. Therefore, in the investment casting process, a 1,000°C mold is adopted to produce castings with the following specifications:
• Material: SAE 310
• Cross section: 2.5 mm
• Casting temperature: 1,700°C
The use of stainless steel is increasingly common in the automotive industry. Since cast stainless steel is already widely used for aircraft parts it is an excellent choice for automotive material conversion. It offers higher strength, can be cast to thinner sections, (weight) reduction) and improves mechanical properties in hightemperature operating conditions.
References 1. Nobuyoshi Sasaki, “A Revolutionary Inorganic Core and Moldmaking Process,” Foundry Management & Technology (Feb. 2009).
2. Nobuyoshi Sasaki, William L. Tordoff, “Thin- Wall Steel Casting Process for Exhaust Components,” Society of Automotive Engineers Inc., 012BECB-43, Paris, France (2002).
3. William L. Tordoff, “Hybrid Casting Process Produces Thin-Walled Ferrous Component,” Modern Casting, November (1999).
4. WTEC (NSF), “Advanced Casting Technology in Japan and Europe,” AFS & International Technology Research Institute (1997).
5. N. Sasaki, “The Development of a Process for Manufacturing Mould for Thin Wall Castings,” BCIRA World Conference Best Paper Awards, York, England (1996). 6. N. Sasaki, “Hollow Castings for Commercial and Automotive Industries an Alternate Cost Effective Solution,” The 9th World Conference on Investment Casting, San Francisco (1996).
7. N. Sasaki, USP5569320 USP5611848 EP066 12 45 B1 1996, Oct. 29; “Process for Preparing Refractory Molded Articles and Binder Therefore” (1996)
8. B. Holmsquist, “Conversion of Quartz to Tridymite,” J. Am. Ceram. Soc. 44[2], (1961)
9. “Selection of Ferrous and Refractory Alloys” and ” Investment Castings for Engineers,” published in 1952 by Rawson L. Wood and David Lee Von Ludwig, and ”Chemical and Heat Resistant Applications,” are quite useful to show the entire specification of steel by high-temperature molding, reflecting a variety of practical experiences during World War II.